The estimates of V2/F, Q/F and V3/F were 22.7 L, 76.3 L/h and 916 L, respectively. The final model including two covariates, population (liver transplant recipients or volunteers) and serum ALT (alanine aminotransferase) level, was verified and adequately described the pharmacokinetic characteristics of tacrolimus. Results:Ī two-compartment model of tacrolimus could best describe the data from the two populations. The final model was validated using bootstrapping and a visual predictive check.
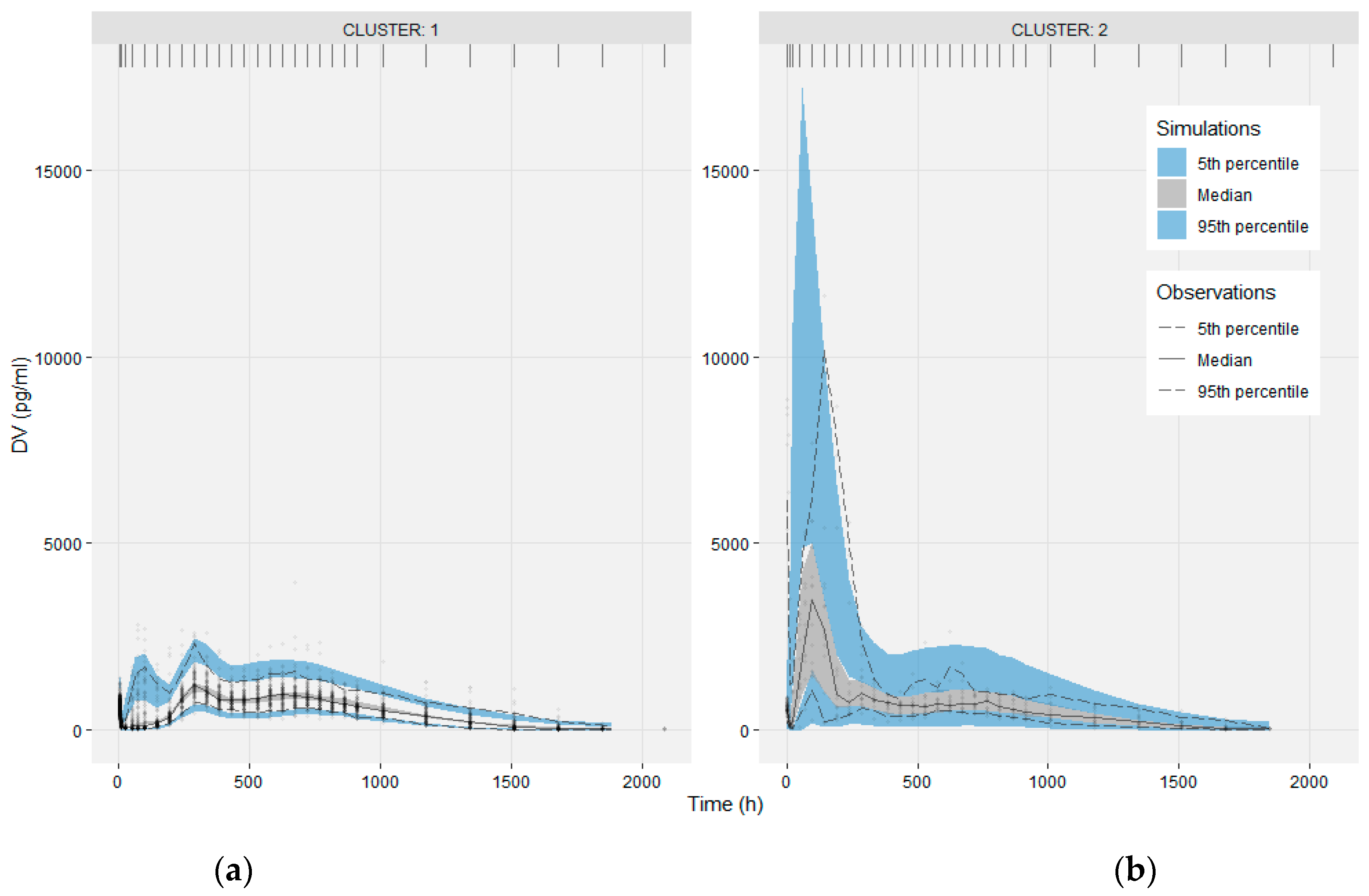
Related covariates such as age, hepatic and renal functions that were potentially associated with tacrolimus disposition were evaluated. PopPK model of tacrolimus was constructed using the program NONMEM. Methods:Ī set of 1100 sparse trough concentration data points from 112 orthotopic liver transplant recipients, as well as 851 dense data points from 40 healthy volunteers receiving a single dose of tacrolimus (2 mg, po) were collected.

Tel.: +1 4 Fax: +1 4 E-mail: Received 18 November 2015 revised 12 March 2016 accepted 7 April 2016 1 2 3 4,5 6 7 Shringi Sharma, Steve Caritis *, Gary Hankins, Menachem Miodovnik ,MaryF.To develop a population pharmacokinetic (PopPK) model of tacrolimus in healthy Chinese volunteers and liver transplant recipients for investigating the difference between the populations, and for potential individualized medication. British Journal of Clinical Pharmacology Wiley īritish Journal of Clinical Br J Clin Pharmacol (2016) 82 1084–1093 1084 Pharmacology DRUGS IN PREGNANCY AND LACTATION Population pharmacokinetics of 17α-hydroxyprogesterone caproate in singleton gestation Correspondence Shringi Sharma, Department of Pharmaceutical Sciences, University of Pittsburgh, Pittsburgh, PA, USA. Tel.: +1 4 Fax: +1 4 E-mail: Received 18 November 2015 revised 12 March 2016 accepted 7 April 2016 1 2 3 4,5 6 7 Shringi Sharma, Steve Caritis *, Gary Hankins, Menachem Miodovnik ,MaryF.Hebert, Don Mattison and Raman Venkataramanan 1 2 Department of Pharmaceutical Sciences, School of Pharmacy, University of Pittsburgh, Pittsburgh, PA, USA, Departments of Obstetrics, Gynecology and Reproductive Sciences, Magee-Women’s Hospital, University of Pittsburgh Medical Center, Pittsburgh, PA, USA, Department of Obstetrics and Gynecology, University of Texas Medical Branch, Galveston, TX, USA, Medstar Health Research Institute, Hyatsville, MD, USA, 5 6 Georgetown-Howards University Center for Clinical and Translational Science, Washington, DC, USA, Department of Pharmacy and Obstetrics & Gynecology, University of Washington, Seattle, WA, USA, and McLaughlin Centre for Population Health Risk Assessment, University of Ottawa, and Risk Sciences International, Ottawa, ON, Canada *Principal Investigator: Steve Caritis Keywords 17α-hydroxyprogesterone caproate, population pharmacokinetics, preterm birth AIMS 17α-hydroxyprogesterone caproate (17-OHPC) reduces

Mattison, Don Venkataramanan, Ramanīritish Journal of Clinical Br J Clin Pharmacol (2016) 82 1084–1093 1084 Pharmacology DRUGS IN PREGNANCY AND LACTATION Population pharmacokinetics of 17α-hydroxyprogesterone caproate in singleton gestation Correspondence Shringi Sharma, Department of Pharmaceutical Sciences, University of Pittsburgh, Pittsburgh, PA, USA. Sharma, Shringi Caritis, Steve Hankins, Gary Miodovnik, Menachem Hebert, Mary F. Population pharmacokinetics of 17α‐hydroxyprogesterone caproate in singleton gestation Population pharmacokinetics of 17α‐hydroxyprogesterone caproate in singleton gestation
